Quick Summary
- Mitochondria generate chemical energy within cells, play a vital role in health, aging and disease
- Mitochondria have own DNA, inherited only from female parent
- New discovery shows how division of mitochondrial DNA is organized within cells
Aging, neurodegenerative disorders and metabolic disease are all linked to mitochondria, structures within our cells that generate chemical energy and maintain their own DNA. In a fundamental discovery with far-reaching implications, scientists at the University of California, Davis, now show how cells control DNA synthesis in mitochondria and couple it to mitochondrial division.
The work is published July 15 in the journal Science.
“This has very profound implications for human disease,” said Jodi Nunnari, professor and chair of molecular and cellular biology at UC Davis and senior author on the paper.
Mitochondria retain their own DNA from the very distant past, when they were a type of bacteria that moved into other cells and never left. All eukaryotic cells — in plants, animals and fungi — contain mitochondria, which allow oxygen-breathing organisms to obtain energy from respiration.
In human cells, mitochondria are elongated, snaking tubes, with hundreds to thousands of copies of their single chromosome dotted around, packaged in a structure called the nucleoid. While the DNA in the cell’s nucleus comes from both parents, your mitochondrial DNA is inherited only from your mother.
While division of DNA in the cell’s nucleus is tightly controlled, synthesis and division of mitochondrial DNA is “a lot more relaxed,” Nunnari said.
How does the cell decide where all the copies of the mitochondrial DNA should go? And how is their division organized, if it is?
Contact points are crucial
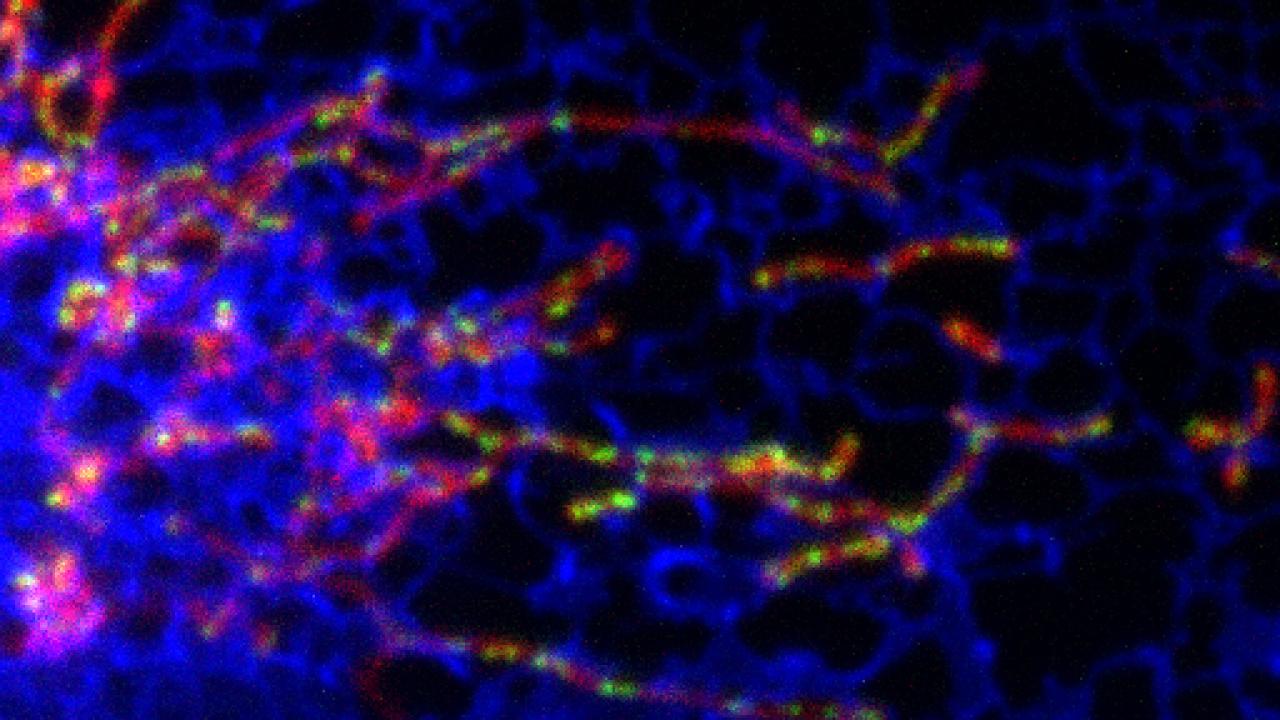
Postdoctoral researcher Samantha Lewis, with undergraduate student Lauren Uchiyama, used microscopy with fluorescent dyes to tag mitochondria, their chromosomes, and the endoplasmic reticulum, a network of tubes that spreads throughout the cell.
They found that dividing mitochondrial chromosomes were located at points where the endoplasmic reticulum touches the outside of a mitochondrion. These also became the points where mitochondria divided into two offspring, a process that requires a sort of lasso of protein around the organelle that squeezes it until it splits.
“The endoplasmic reticulum comes into contact with the mitochondrion, and where they contact is where they divide,” Nunnari said.
The contact between the two organelles “licenses” the mitochondrial DNA to copy and divide, Nunnari said. This DNA division is in turn spatially coupled to division of the mitochondrion itself, and to distribution of the daughter DNA around the cell.
“There are hundreds of contact points around the cell that determine where division takes place and how mitochondria are distributed, but division preferentially occurs at the subset of contacts where mitochondrial DNA is being copied” Nunnari said. “It shows that there is a higher order to this, it is not simply random.”
The discovery has broad implications for understanding cell functions, aging and a broad range of diseases. Nunnari noted that it stemmed entirely from fundamental research.
“We didn’t come to this by studying any specific disease, it’s discovery-based research,” she said. “But this will greatly impact human health.”
The work was funded by the National Institute of General Medical Sciences, part of the National Institutes of Health.
Media Resources
Jodi Nunnari, Molecular and Cellular Biology, 530-754-9774, jmnunnari@ucdavis.edu
Andy Fell, News and Media Relations, 530-752-4533, ahfell@ucdavis.edu