WHEN BRIAN BIRD AND HIS TEAM from the Centers for Disease Control arrived at the hospital in Kenema, Sierra Leone, during the 2014 Ebola outbreak, half the hospital’s staff were dead or missing from work.
“Ebola outbreaks have the singular ability to reveal how fragile our systems are,” Bird said. “We depend on people caring for each other, and Ebola preys on this because it puts caregivers at extreme risk.”
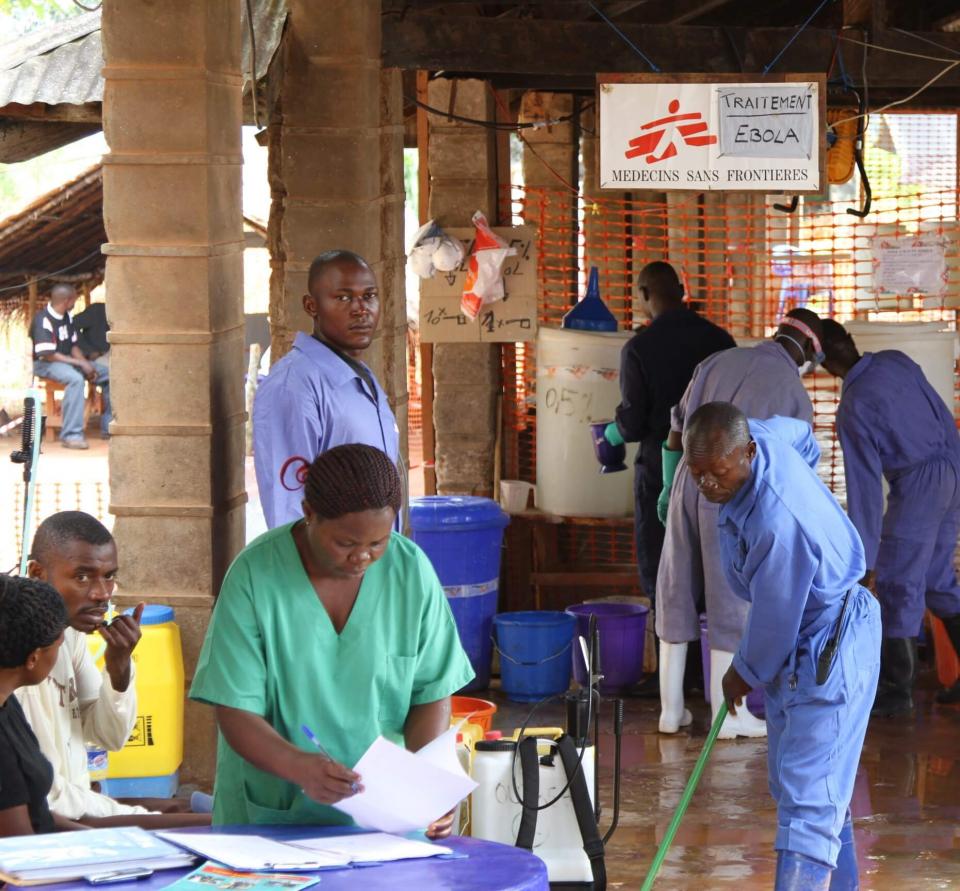
Bird, a graduate of the University of California, Davis, School of Veterinary Medicine, helped coordinate operations of the U.S. government’s diagnostic labs in the region during the outbreak, which sickened at least 28,000 people and killed 11,310. Working alongside colleagues from the CDC, World Health Organization and the medical charity Doctors Without Borders, Bird’s team was responsible for testing patients admitted to the hospital for Ebola, as well as surveillance for new cases in the community. A misdiagnosis could put a patient with some other disease in a ward where they could be exposed to Ebola. Or it could release an Ebola-infected person into the community.
“Ebola outbreaks reveal how fragile our systems are.”
“It was a very challenging and difficult environment because the stakes are so high — life or death for people and communities,” Bird said.
Today, Bird is a member of the PREDICT project, funded by the U.S. Agency for International Development and led by the One Health Institute at the UC Davis School of Veterinary Medicine. The aim of PREDICT is to enable global surveillance for viruses with the potential to spillover from animals to people and cause pandemics.
The hunt for Ebola’s source
Among other duties, Bird now leads PREDICT’s Ebola Host Project, surveying wildlife in Sierra Leone, Guinea and Liberia. Several times in the past 42 years, Ebola has appeared from the forests, started an outbreak of disease in humans and then vanished. Researchers believe that between outbreaks the virus must be in a wild animal host without causing much harm. But this host has remained elusive.


“We still don’t know where the virus comes from. If we knew, we could tell people what to avoid,” Bird said.
Ebola outbreaks are rare, so infected hosts are likely also rare, he added. That means collecting tens of thousands of samples to have a chance of finding the virus.
“We need a big net to catch a rare fish,” he said.
Bird was previously part of the CDC team that identified a species of fruit bat as the natural host of Marburg virus and bats are a possible host candidate for Ebola. In July 2018, the Sierra Leone government announced the discovery of a new species of Ebola virus in bats by a team including Tracey Goldstein, associate director of the One Health Institute, and colleagues at UC Davis and Columbia University. If this particular virus can infect humans or if the Ebola viruses known to cause disease in humans are also hosted by bats is not yet clear.


Training and capacity building
At its core, the search for the hosts of Ebola viruses depends on building up local expertise. When Bird worked on the 2014 outbreak in Sierra Leone, no local teams were trained to perform wildlife surveys. Now, his team is educating forest guides in animal capture and sampling, field laboratory techniques and managing a refrigerated “cold chain” for samples — for an effort roughly 10 times the size of any previous search for the Ebola host.
Training people and building local capacity to detect known and emerging diseases before they can become a threat is a primary goal for PREDICT. In the past eight and a half years, the USAID-funded program has detected more than 1,000 viruses that need further examination to understand if they could emerge as threats to human health, and trained more than 4,200 people in 30 countries to carry out surveillance for viruses.
“When PREDICT first starts working in a country, we visit and ask with whom — government departments, educational and scientific institutions, nonprofits — we can partner,” said Professor Jonna Mazet, executive director of PREDICT and of the One Health Institute. Those trained by the partnership with PREDICT can then train others.

Emerging diseases
Viruses circulate naturally among Earth’s millions of species. Some cause disease, but many are not pathogens: The animal host and the virus have long since learned to coexist.
Sometimes, viruses will jump to another species. That might happen when domestic animals mix with wild ones, or when someone is bitten, scratched, or exposed to feces or body fluids from an infected wild animal. If the virus can adapt to a new host, it may be able to cause infection and in some cases disease in the new host. This kind of infection is called a zoonosis and results in a spillover into humans from an animal.
Such infections are often dead ends for the virus and are not able to transmit from human to human. But with repeated infections and the process of genetic mutation and change, some viruses may acquire the ability to spread from person to person. And a potential epidemic is born.
PREDICT began in 2009 with the goal of understanding viruses before they emerge as pathogens in humans. Following up on its success, the Global Virome Project, announced in February 2018 and also led by Mazet’s team at UC Davis, aims to identify up to a million viruses in wildlife and to develop tools that enable scientists and policymakers to identify hotspots where new diseases are most likely to emerge.

The urban interface
Those hotspots could be tropical forests that are opening up to human settlement. Or they could be urban slums, where people live with poor sanitation and health care in close contact with domestic animals and wild rodents.
Since 2011, the PREDICT project has been working with communities around Kathmandu, the capital city of Nepal. In temporary, unplanned settlements, swelled by the devastating earthquake of 2015, impoverished people live along rivers, raising ducks and pigs and growing rice and vegetables alongside wild rodents and other wildlife.
“These areas have all the ingredients for emerging diseases.”
“These areas have all the ingredients for emerging diseases,” said Professor Christine Johnson of the One Health Institute. The rats, mice and shrews found in rice fields and people’s homes are top of the list for spillover of viruses from the wild to the human population.
PREDICT‘s implementing partner, the Center for Molecular Dynamics Nepal, has been working with communities and government partners to sample the people, their livestock and the wild animals in their fields and homes.
Nonhuman primates are a particular threat for disease transmission because they are so closely related to us. Macaque monkeys thrive in South Asian cities and are especially common at religious shrines, “monkey temples” where tourists and worshippers feed the animals. The monkeys are revered by the Nepalese and therefore protected from harm — although that doesn’t stop fairly frequent monkey bites, Johnson said.
To sample the monkeys while respecting local culture, UC Davis postdoctoral scholar Tierra Smiley Evans came up with a simple approach: a short piece of rope dipped in strawberry jam or banana paste. The monkeys pick up the rope, chew on it and discard it, leaving a nice sample of monkey saliva. After testing this approach with macaques at the California National Primate Research Center, and with wild primates in Uganda and the help of the Mountain Gorilla Veterinary Program (affiliated with the UC Davis School of Veterinary Medicine), the PREDICT surveillance team in Nepal has used this technique to identify viruses in primate species.

Smartphones and community health in the Philippines
Community health workers trained in a “One Health” approach — that encompasses not just the health of people but also of their domestic animals, environment and even crops — could provide a valuable service for rural communities. These community health workers are on the front line to identify, isolate and treat patients during a disease outbreak.
On the island of Luzon in the Philippines, Professor Michael Wilkes of the UC Davis School of Medicine is leading an ambitious project, named the Philippine-California Advanced Research Institutes (PCARI) Project, funded mostly by the Philippine government, to train such One Health workers. Their key tool? A smartphone app.
Health care may be rudimentary in the rural Philippines, but cellphones are inexpensive and widely used, Wilkes said.
The app guides health workers through a series of questions and provides guidance, such as whether a patient should be treated on the spot or if the case should be referred to a district health center, hospital or veterinary center. When the patient arrives at the hospital, information gathered through the app is immediately available to physicians.
Every night, data collected on the One Health workers’ phones is uploaded to a central server. There, broader patterns emerge: for example, an outbreak of Newcastle disease in chickens in several villages, or the beginnings of a human influenza epidemic.
“It’s a whole new way of looking at disease identification, based on community involvement,” Wilkes said.
The project is currently in the second of four years. If it is successful, Wilkes hopes that it could be transferred to other developing countries with basic health care but ubiquitous cellphones.

Medical innovations: Flu sniffers and bioreactors
Other technology developed at UC Davis could help fight off global pandemics. Professor Cristina Davis, of the Department of Mechanical and Aerospace Engineering, is working on a device to detect influenza, based on a patient’s breath. With grant support from the Defense Advanced Research Projects Agency and the National Institutes of Health, Davis has developed techniques to detect very low levels of chemicals in odors for a variety of applications in health, agriculture and research. She is working with professors Mike Schivo and Nick Kenyon at UC Davis Health and Professor Susan Ebeler at UC Davis’ Department of Viticulture and Enology to refine the “flu sniffer.” It has proved successful in tests with cultures of flu-infected cells and is currently being tested in patients.
As the 2014 Ebola outbreak was raging in West Africa, UC Davis engineering professor Karen McDonald received a “rapid response” grant from the National Science Foundation. The task: To develop a platform for rapid production of biologics such as the promising anti-Ebola drug ZMapp (developed by Mapp Biopharmaceutical of San Diego and collaborators in Canada) in plant cells in a bioreactor.
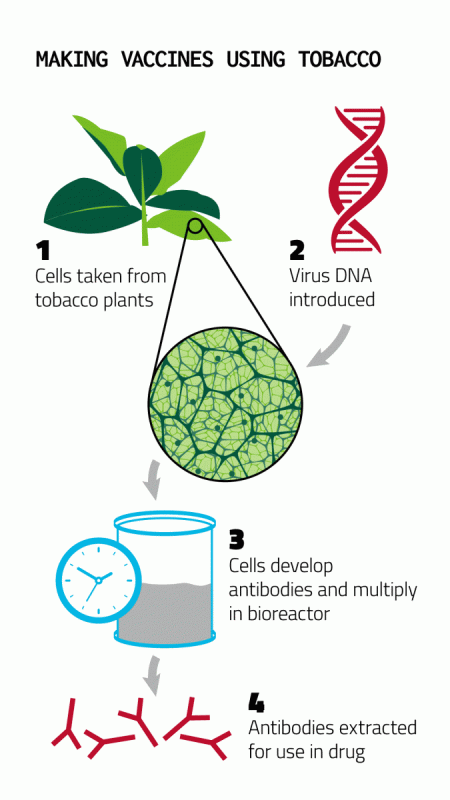
At that time, ZMapp was being produced in tobacco plants that were then ground up to release the drug. McDonald’s approach was to grow cultures of tobacco plant cells in bioreactors used by the biotechnology industry and develop a system to deliver genes for the product of interest to the plant cells. If successful, this approach should allow rapid production of therapeutics in existing biomanufacturing facilities. McDonald’s lab continues to work on optimizing the process, which could be applied to other drugs needed for emerging infectious diseases or biothreats.
The team is also implementing single-use bioreactors for plant cell cultures that are robust and easy to use in areas with few resources. Such systems could be used to produce drugs and vaccines in bulk close to the point of need, rather than having to ship perishable medicines long distances into the field.


Training for the future
Heading off future pandemics will require collaboration among physicians, veterinarians, scientists and public health experts who are grounded in the complexities of disease in a changing world. Two UC Davis programs aim to address that need.
RX One Health is a monthlong summer program run by a collaboration between UC Davis’ One Health Institute, School of Medicine and College of Agricultural and Environmental Sciences, together with the University of Rwanda and Sokoine University of Tanzania. Now in its third year, RX One Health brings medical, veterinary and doctoral students to work and study with local students in developing countries, building both their own skills and local expertise. Participants meet with physicians, farmers, veterinarians, midwives, government officials, and other local health and science professionals.
“They learn to work as teams, veterinarians with physicians and agricultural scientists, on the same problems,” said Wilkes, one of the program’s organizers.
This on-the-ground experience can bring some fresh insights. For example, when UC Davis veterinary student Taylor Calloway visited a Tanzanian fishing village, she assumed the villagers would need clean water and better housing. Instead, the locals’ main concerns were attacks by hippos and crocodiles that prevented them working.
At the undergraduate level, UC Davis’ major in global disease biology exposes students to the complexities of health care in a dynamic and complex world. David Rizzo, professor of plant pathology who helped spearhead the major in 2014, described it as a “super-sized public health major.”
The major capitalizes on the unique opportunities present on the UC Davis campus. Through transdisciplinary courses taught by professors in different departments across the schools of medicine and veterinary medicine and the College of Agricultural and Environmental Sciences, students learn about changing environments, infectious diseases, health policy and different cultures — and how these seemingly unrelated threads intertwine in a global health network.
“In order to solve some of these really big picture health issues, we can’t just consider human health,” said Rizzo.
Students complete a senior research project that allows them to bridge disciplines, receive faculty mentorship, and get behind the lab bench or out in the field. Gaining tangible experience augments their understanding of disease.
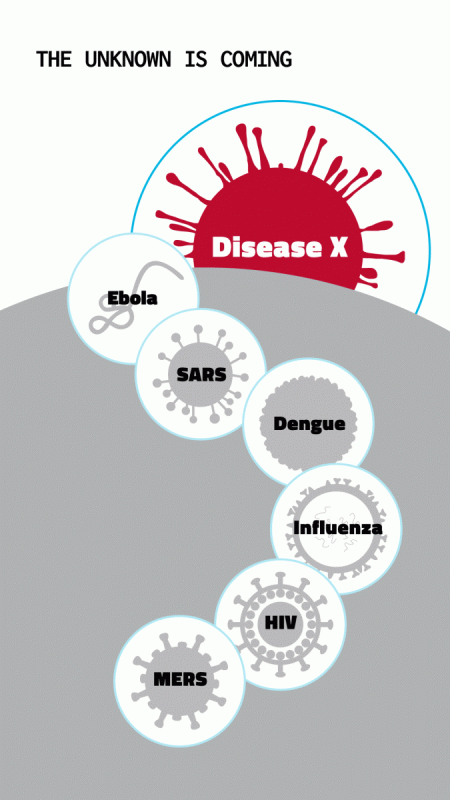
In February 2018, the World Health Organization updated its Blueprint list of diseases that should be a priority for accelerated research and development. These are diseases that experts judge could cause a public health emergency but lack effective vaccines or treatments.
Among those on the list were Ebola and Marburg viruses, Middle East respiratory syndrome (MERS) virus and SARS virus, Zika virus — and “Disease X,” representing “the knowledge that a serious international epidemic could be caused by a pathogen currently unknown to cause human disease,” according to WHO.
UC Davis is a leader in training a new generation of health workers and is continually collaborating and innovating to find and stop future pandemics.

Media contact: Andy Fell, UC Davis News and Media Relations, (530) 752-4533, ahfell@ucdavis.edu
Related Stories
Healing Burned Animals with Fish Skins
Cinders was the first horse to be treated with tilapia skins for burn wounds, but the first animal test case came in October 2017 during the Thomas Fire in California’s Ventura and Santa Barbara counties.

Guardians of the Genome
One in eight women will be diagnosed with breast cancer at some point in their lives, but through new discoveries at the genetic level the personal nature of cancer will eventually be what helps to beat it.
Minibrains Move a Step Closer to Helping Patients
UC Davis brain surgeon Ben Waldau has groundbreaking success growing parts of human brains.
